  
This document is available for review at the Department of Commerce Library, Main Commerce Building, Washington, D.C., under reference number BAW-1130.Trending Questions Which school of thought held that the universe is governed by gods who were not interested in people and the only real objects are those that can be perceived by the five senses? Why is it necessary to use special instruments to measure the temperature of the atmosphere in the thermosphere layer? What appears in a dark circle inthe night sky on certain days? Which of these sciences is not a natural science astronomy anthropology or zoology? Who was the man who slept for 18000 years then created the universe was? Which member of the solar system has an equatorial diameter of 3.48 times 10 to the 3rd? Is it possible to have total internal reflection from light incident from air on water? What are some products named after Astronomical objects? What is the energy of an asteroid? What are common phrases that imply an Earth centered viewpoint? If it is noon at one location what time is it on the other side of the earth? How long will it take you to reach Alpha Centauri 4.37 light years away if you are travelling 16000 mph? Is a satellite circling the Earth above the atmosphere in free fall? What is so special about c? Which was the first company in the world to build computer for sale? The time at which the Moon rises changes by about 50 minutes every day.How much of a 1.000 mg sample will remain after 15.9 years Dating Because the half-life of any nuclide is constant, the amount of substance remaining in an artifact can serve as a nuclear clock to determine ages of objects. Their accuracy is limited, of course, by the validity of the assumptions used in setting up and solving the equations, and by the accuracy of the evaluation of the constants. Example: The half-life of cobalt-60 is 5.3 years. Calculations were then made for the Nuclear Merchant Ship Reactor. Solutions were found for the equations and the constants evaluated from APPR-1 data. The approach was to derive a set of differential equations which should describe the activity buildup phenomenon. Accordingly, the object of this investigation was to determine the concentration of Cobalt allowable as an impurity in the steel which would give no greater activity from Cobalt-60 than from Cobalt-58. Practically, there is no object in reducing the Cobalt impurity to a point where the Cobalt-60 activity becomes less than the Cobalt-58 activity. Cobalt-60 Co CID 61492 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards. 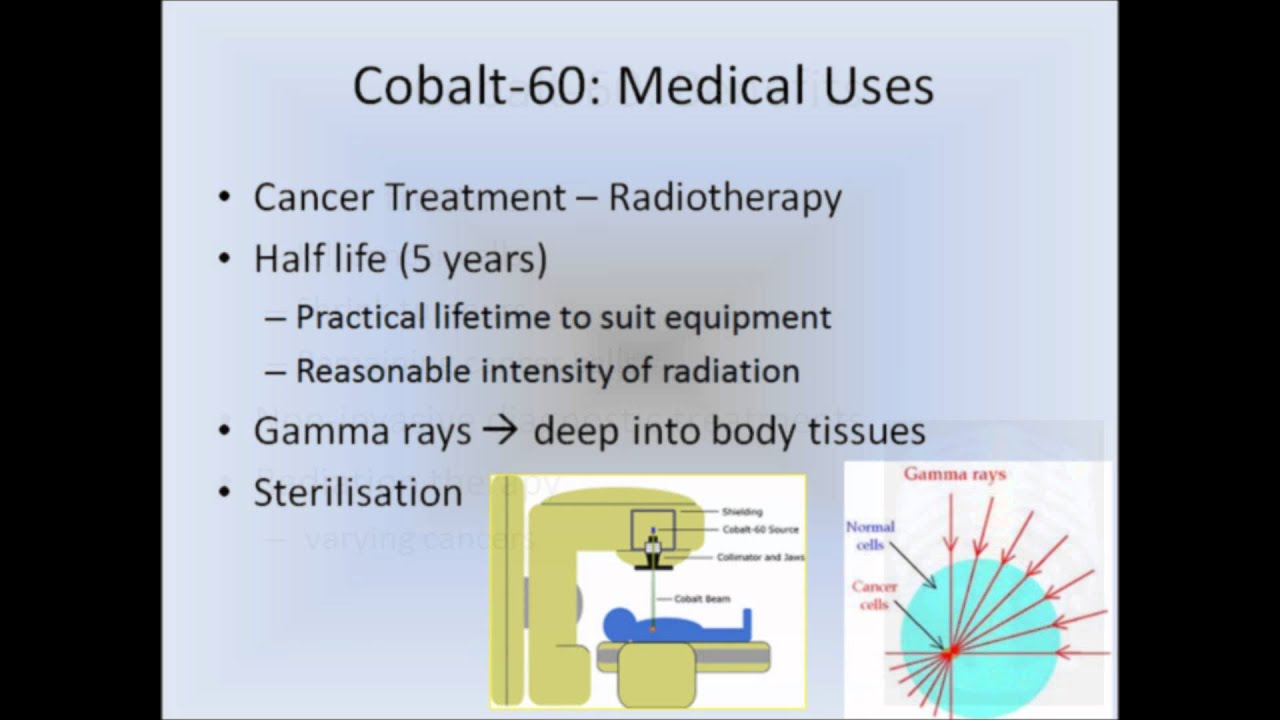
Accordingly, there is some economical limit on the degree of reduction. 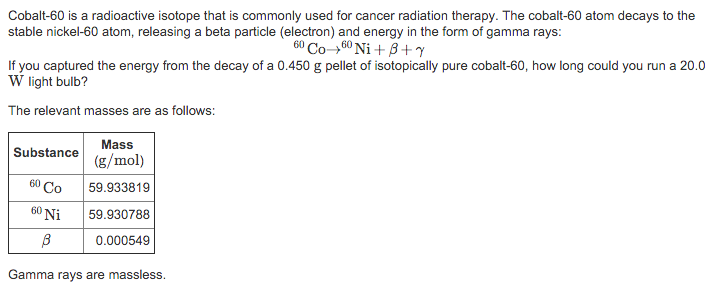
The greater the reduction in the cobalt impurity, the more expensive the steel becomes. Write the decay equation for an excited atom of Cobalt-60 undergoing gamma decay. If this is reduced to 0.01%, the Cobalt-60 buildup will be reduced accordingly. Sum of superscripts : 211 y + 0, so y 211. The amount of Cobalt in 304 stainless steel varies considerably but is generally on the order of 0.1%. The main point to remember in balancing nuclear equations is that the sums of the superscripts and the subscripts must be the same on each side of the equation. However, the Cobalt (which is 100% Cobalt-59) is present as an impurity, and it is possible to reduce its concentration considerably. In a material such as 304 stainless steel, it is obvious that the nickel cannot be eliminated. The cobalt-60 atom decays to the stable nickel-60 atom, releasing a beta particle (electron) and energy in the form of gamma rays: 60Co 60Ni + +. Cobalt-58 is formed from the n,p reaction of Nickel-58 while Cobalt-60 is formed from thermal neutron absorption by Cobalt-59. Two of the most active radioisotopes in the deposit are Cobalt-58 and Cobalt-60. This activity buildup is particularly bothersome during maintenance, i.e. In a pressurized water reactor constructed with stainless steel such as the Nuclear Merchant Ship Reactor, a radioactive deposit (commonly called crud) tends to buildup on the walls of the primary system. This type of nuclear reaction is called A)nuclear fusion B)nuclear fission C)natural transmutation D)artificial transmutation 20. The gamma spectrum has two significant peaks, one at 1173.2 keV and another at 1332.5 keV. Cobalt-60 is a common calibration source found in many laboratories. It is synthetically produced by neutron activation of cobalt-59 in nuclear reactors. That property does have positive uses (for things like medical x-. Cobalt-60 is an artificial radioactive isotope of cobalt with a half-life of 5.2747 years. Cobalt-60, which is the most common nuclide used in radiation therapy for cancer. COBALT ACTIVITY BUILDUP IN THE NUCLEAR MERCHANT SHIP REACTOR D)nuclear fission 19.Radioactive cobalt-60 is used in radiation therapy treatment. Cobalt-60 decays (with a half life of about 5 years) by emitting a very high energy gamma ray. To show how nuclear equations are different from chemical equations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
